Membrane protein
Membrane (M) protein is the most abundant of all coronavirus structural proteins. M protein main functions are membrane curvature initiation, RNA packing and viral particle budding. Besides, M protein has been linked to apoptosis, suppression of inflammatory responses and IFN signaling. Most of the variation between SARS-CoV and SARS-CoV-2 M occurs in the cytoplasmic domain at the C-terminus, involving several sites that can potentially undergo post-translational modification. Therefore, these mutations could affect M protein function and antigenic properties.
Narrative
Membrane (M) proteins are the most abundant of all coronavirus structural proteins, and their presence and conformation settle the virion shape (K. Narayanan and Makino 2001; Lili Kuo and Masters 2002). M protein has been shown to interact with viral proteins N, S, ORF3a and ORF7a (Y. Huang et al. 2004; He, Dobie, et al. 2004; Tan et al. 2004; Fielding et al. 2006). The general structure of M proteins are that they contain a short N-terminal ectodomain, three transmembrane domains (TMD), and a C-terminal endodomain (Armstrong et al. 1984). M proteins may work as homodimers that can take compact and long conformations (Neuman et al. 2011). The long conformation is associated with high S protein density and efficient virus budding, while compact conformation is associated with patchy and low S protein presence and inefficient budding (Neuman et al. 2011). Only M and E proteins are required to form virus-like particles (VLP), but absence of S proteins makes VLPs appear larger, suggesting they may contribute to virus formation (Neuman et al. 2011). SARS-CoV-1 M protein has been shown to interact with N protein in mammalian two-hybrid assays (He, Leeson, et al. 2004). Only RNAs interacting with M protein can be found in viral particles in mouse hepatitis virus (MHV) (K. Narayanan and Makino 2001).
The M protein has been demonstrated to be a pathogenic factor in coronaviruses. For instance, overexpression of M protein in vitro and in vivo induces apoptosis in human cell culture and Drosophila, respectively (Chan et al. 2007). Further, SARS-CoV-1 M protein was found to interact with IKKβ, impairing NF-κB signaling and reducing the expression of cyclooxygenase-2 (COX-2) (X. Fang et al. 2007). COX-2 is responsible for the production of prostaglandins, which in turn trigger inflammatory responses. Furthermore, interferon (INF) are a group of antiviral proteins produced by cells in response to a viral infection. M protein impairs the production of type-I INF by interfering with TRAF3-TANK-TBK1/IKKε complex formation, thus preventing IRF3 phosphorylation (Siu et al. 2009). Nonetheless, antigenic regions of M protein are detected and production of INF is triggered (Laude et al. 1992). These antigenic properties were observed in patients after a year of recovery from SARS-CoV-1 infection, showing persistent IFNγ release from CD4+ and CD8+ lymphocytes when cocultured with M peptides (L. Yang et al. 2007). Thus, SARS-CoV-1 infection triggered the production of memory cells and M protein could be an interesting vaccine candidate.
Structural analysis and comparison with SARS-CoV-1 membrane protein – Several key functional residues have been identified for the M protein. Transmissible gastroenteritis virus (TEGV) showed reduced IFN induction when mutations occur in the N-terminal residues 6 – 22 (Laude et al. 1992). Alteration of the Asn-Ser-Thr motif in this region prevented glycosylation of the M protein, suggesting its importance in INF induction (Laude et al. 1992). In another study, after several subculture passages in immortalized proximal tubular epithelial cells (PTEC), the SARS-CoV-1 M protein generated a glutamic acid to alanine point mutation at position 11 (Pacciarini et al. 2008). This mutation enhances virus replication and persistence in PTEC (Pacciarini et al. 2008). Tseng et al.(2013) identified other key residues in the SARS-CoV-1 M protein with different functions in replication. The Leu-Leu motif (Leu218 and Leu219) is required to incorporate the N protein into VLPs. Substitution Cys158Ser reduces secretion of the M protein, but not Cys63Ser or Cys85Ser, and Cys158 is involved in the interaction with the N protein. Non conservative substitutions in the motif Ser-Trp-Trp-Ser-Phe-Asn-Pro-Glu reduced the production of VLPs. Conversely, substitutions of the aromatic residues in this motif by analogs did not affect the VLPs production, suggesting that the conservation of these aromatic amino acids is critical for M protein function.
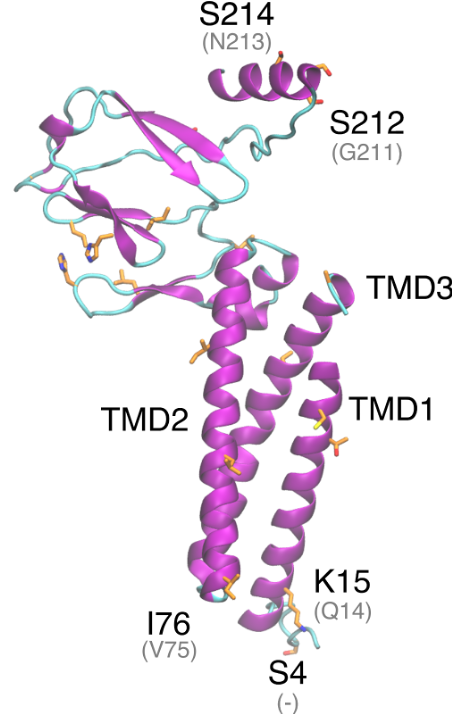
The SARS-CoV-2 M protein is predicted to have three transmembrane domains, a cytoplasmic and a non-cytoplasmic domain. This is similar to the SARS-CoV-1 M protein and other coronavirus M proteins (Armstrong et al. 1984; Y. Hu et al. 2003). InterProScan (Jones et al. 2014) defined these regions as follows: non-cytoplasmic region (1 – 19), TMD1 (20 – 40), cytoplasmic region (41 – 51), TMD2 (52 – 73), non-cytoplasmic region (74 – 78), TMD3 (79 – 103) and cytoplasmic region (103 – 222). The SARS-CoV and SARS-CoV-2 M proteins are very similar, with 90·54% identity. Among 21 variations, 12 are non-conservative substitutions and one is an insertion. Within the substitutions, Gln15Lys may have an effect in antigenic properties of the M protein, by modifying its ectodomain. Besides, Ser4 insertion is very close to a predicted N-glycosylation site at the N-terminal, and can constitute an additional glycosylation site. Modifications to this glycosylation site have been associated with induction of INF production (Laude et al. 1992). Most of the non-conservative mutations occur in the cytoplasmic domain at the C-terminal domain. These substitutions involve several sites that can potentially undergo post-translational modification (Ser155His, Thr189Gly, Asn197Ser, Ala211Ser, Gly212Ser and Asn214Ser).
